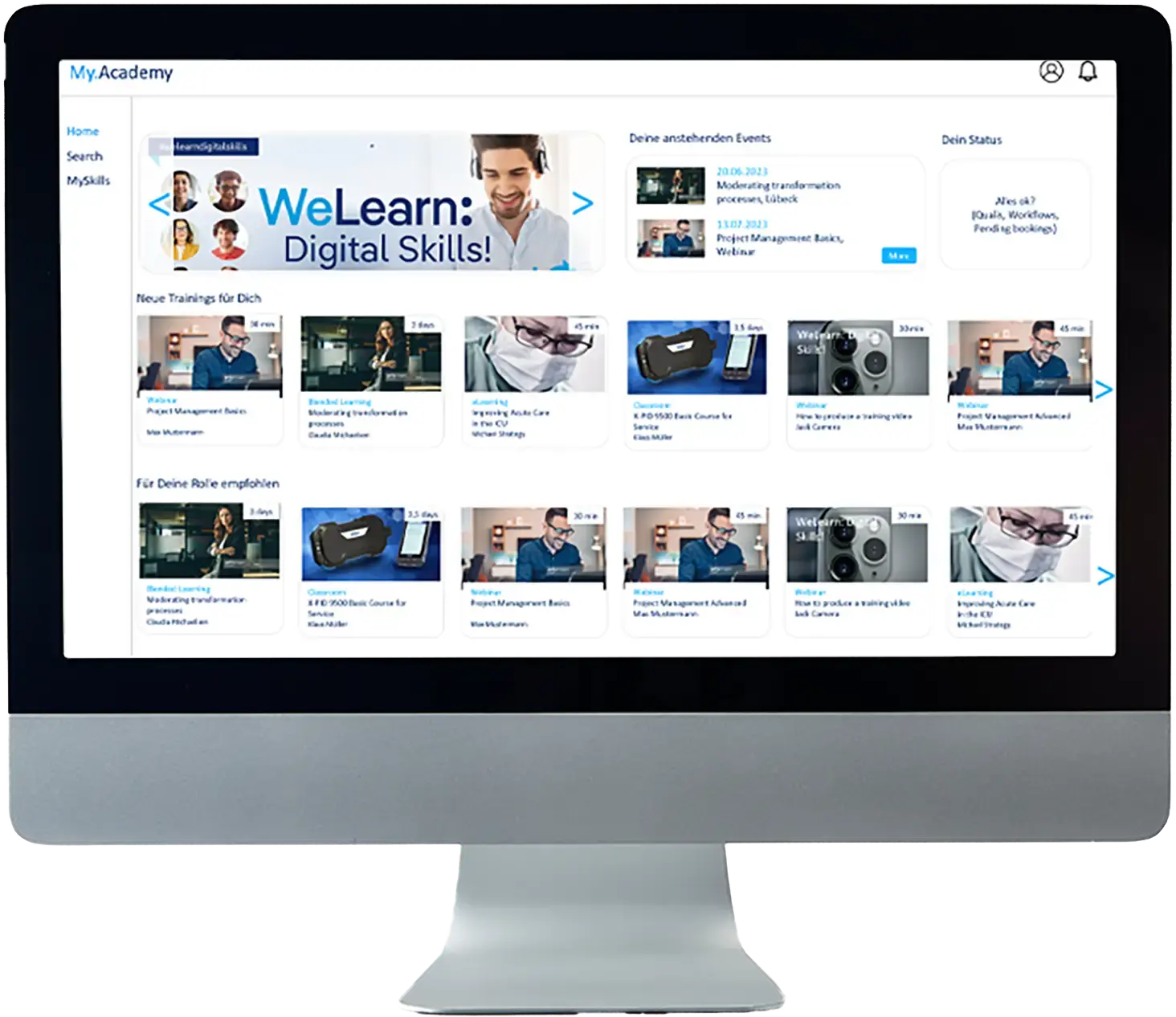
Meet Cmpliance Trainign Requirements – TCmanager® LMS
In regulated industries, compliance with legal requirements and standards such as ISO, GDPR, FDA, or OSHA is essential.
The TCmanager® Learning Management System helps organizations efficiently organize, document, and provide verifiable compliance training records for audits.
Automate mandatory training, manage qualifications transparently, and provide audit evidence at any time: TCmanager® is a specialized LMS for compliance training designed for organizations with high regulatory requirements. From rule-based planning to audit-proof documentation, it covers the entire compliance training lifecycle - ideal for companies operating in highly regulated environments.
Learning Management System - Demovideo




Why do you need an LMS for compliance training?
Compliance requirements are becoming more complex. Tighter deadlines, more detailed audits, and stricter documentation obligations quickly overwhelm Excel spreadsheets and manual processes.
Typical challenges include:
- Managing mandatory training in spreadsheets
- Lack of visibility into deadlines and qualification gaps
- High administrative effort for reminders and audit preparation
A dedicated LMS for compliance training centralizes and automates these processes, increases legal certainty, and significantly reduces the workload for HR and specialist departments.
TCmanager® - your LMS for Compliance Training
TCmanager® is not any generic learning management system. It is purpose-built for audit readiness, structured qualification management, and regulatory requirements such as data protection and ISO standards. Compliance training is managed systematically, transparently, and at scale.
| Compliance training requirement | How TCmanager® supports it | Value for your organization |
| Managing mandatory training | Rule-based assignment by role and location |
The right training at the right time |
| Meeting deadlines | Automated reminders and escalations | No missed deadlines |
| Providing audit evidence | Audit-proof reports and histories | Confidence during audits |
| Demonstrating qualifications | Linked to role-based profiles | Transparent proof of competence |
| Combining learning formats | E-learning, classroom, webinar, blended | Consistent tracking across formats |




Automated assignment of mandatory training
TCmanager® assigns mandatory training based on clearly defined rules—for employees, external staff, or teams, depending on role and location.
Key features:
- Target-group-specific workflows
- Real-time visibility into deadlines and deviations
- Automated recertification cycles
This significantly reduces manual effort and ensures deadlines are reliably met.
Audit-ready reports for inspections
TCmanager® provides audit-proof documentation of all relevant data, including participation, test results, certificates, and training histories.
Available reports include:
- Open or overdue training by department
- Qualification status at team or location level
- Export-ready documentation for auditors
This minimizes audit preparation effort and increases confidence during internal and external reviews.
Integrated Qualification Management
TCmanager® goes beyond basic course administration. Role profiles define which mandatory trainings and certifications are required, while dashboards highlight qualification gaps early: at individual, team, or cross-location level.

Flexible learning formats and certification
TCmanager® consistently tracks all common compliance training formats, including e-learning, classroom training, webinars, and blended learning programs.
Designed for compliance requirements:
- Integrated tests and final assessments
- Automatic certificates with validity periods
- Seamless tracking across all learning formats
Seamless integration into HR processes
Interfaces to HR systems ensure that employee master data remains up to date. Compliance rules are applied automatically when new employees join or when roles change.
Role-specific reports provide relevant insights for HR, compliance teams, and management.
Compliance LMS for Regulated Organizations
TCmanager® is particularly well suited for organizations with:
- Multiple locations or international teams
- Strongly regulated industries such as manufacturing, finance, or healthcare
- Mandatory training requirements in occupational safety and data protection





IT Security & Regulatory Compliance
Developed & hosted in Germany: TCmanager® is operated in ISO-certified data centers, fully compliant with GDPR requirements, and offers granular role and permission management.
This makes it well suited for training programs related to data protection, ISO standards, and information security, combined with transparent qualification tracking.
FAQs: Compliance with LMS - Your Most Frequent Questions
How can I ensure that all employees are trained?
Through automated reminders, individual learning paths, and transparent progress reports.
Are the records accepted by authorities and auditors?
Yes, TCmanager® stores all records in an audit-proof and ISO-compliant manner, exportable for auditors.
Can external staff and partners be included?
Yes, via individual learning portals with secure access, role-based permissions, and data protection.
Does the system meet data protection and IT security requirements?
Absolutely. TCmanager® is GDPR-compliant and ISO 27001-certified, ensuring full data protection.




Schedule Your Free Consultation
Get personalized advice on the optimal implementation of your compliance and audit requirements.
Next steps:
- Schedule a live demo
- Review integration options
- Discuss industry-specific use cases
📞 Tel.: +49 (0)89 / 3090 839 30
.jpg)